Acetic Acid To Acetic Anhydride
Acetic Acid To Acetic Anhydride. It is widely used as an acetylating agent or in chemical synthesis. Acetic anhydride acetic acid anhydride ethanoic anhydride cas no.
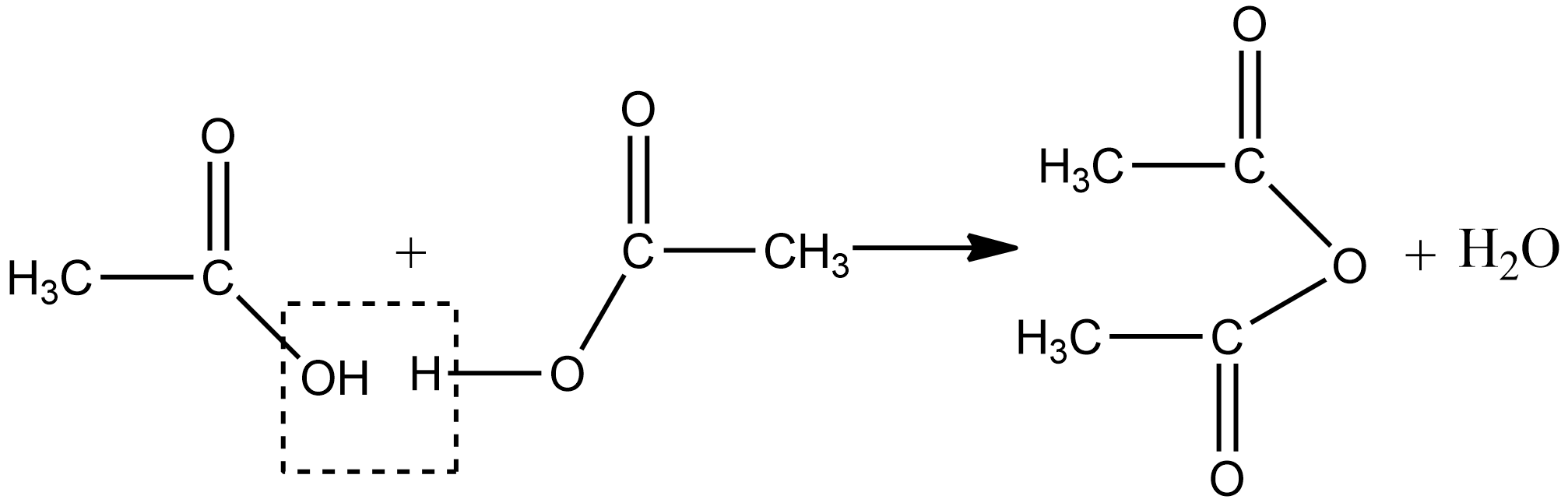
 Acetic anhydride is prepared in the laboratory by heating from www.vedantu.com
Acetic anhydride is prepared in the laboratory by heating from www.vedantu.comThe thermal cleavage of acetic acid to form ketene and the reaction of ketene with acetic acid. Acetic acid is an organic compound having the chemical formula ch3cooh, while acetic anhydride is an organic compound having the chemical formula (ch3co)2o. Acetic anhydride is a carboxylic acid anhydride commonly used for the acetylation of amines and alcohols.
As the defining ingredient of vinegar, acetic acid has been produced and used by humans since before the dawn of recorded history. Warm the mixture on water bath at 60 degrees about 20 min with frequent stirring.

Often used as a monomer for acrylic and vinyl acetate monomers and in polyimide and polyimide precursor synthesis, and resin and hardener production. Copy sheet of paper on top of another sheet.
Being a carboxylic acid anhydride, acetic anhydride will slowly decay in contact with waterinto acetic acid. This process yields aspirin and acetic acid, which is considered a byproduct of this reaction.
It is widely used as an acetylating agent or in chemical synthesis. Warm the mixture on water bath at 60 degrees about 20 min with frequent stirring.
We are the 7th largest manufacturer of acetic anhydride in the world and are 4th largest in terms of merchant sales globally. Acetic acid is an organic compound that is very useful in different organic synthesis reactions.

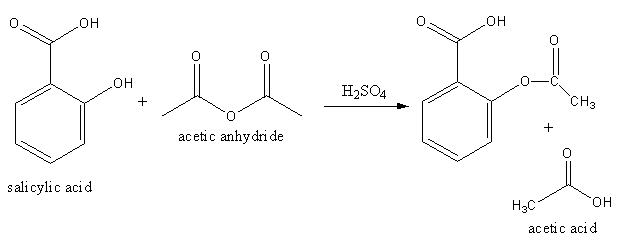
The key difference between acetic acid and acetic anhydride is that acetic acid is a simple carboxylic acid, whereas acetic anhydride is the dehydration product of acetic acid. Salicylic acid acetic anhydride acetylsalicylic acid (aspirin) acetic acid preparation of acetylsalicylic acid.
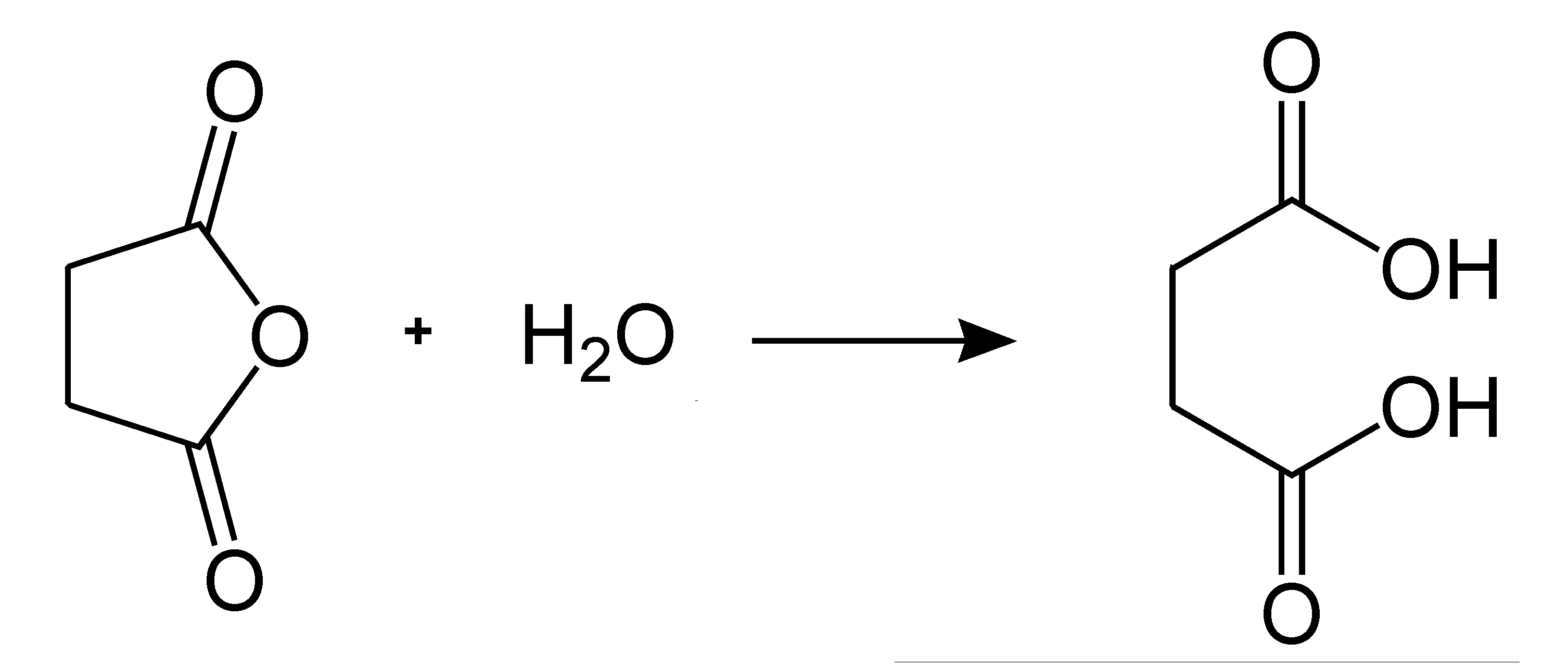
The thermal cleavage of acetic acid to form ketene and the reaction of ketene with acetic acid. With pcl5, the reaction is a two step process.

Salicylic acid acetic anhydride acetylsalicylic acid (aspirin) acetic acid preparation of acetylsalicylic acid. Each unique type of carbon atom in a molecule will give a different peak in the spectrum.
The largest volume use for acetic anhydride is as a raw material for cellulose acetate fibers and plastics. Acetic anhydride acetic acid anhydride ethanoic anhydride cas no.
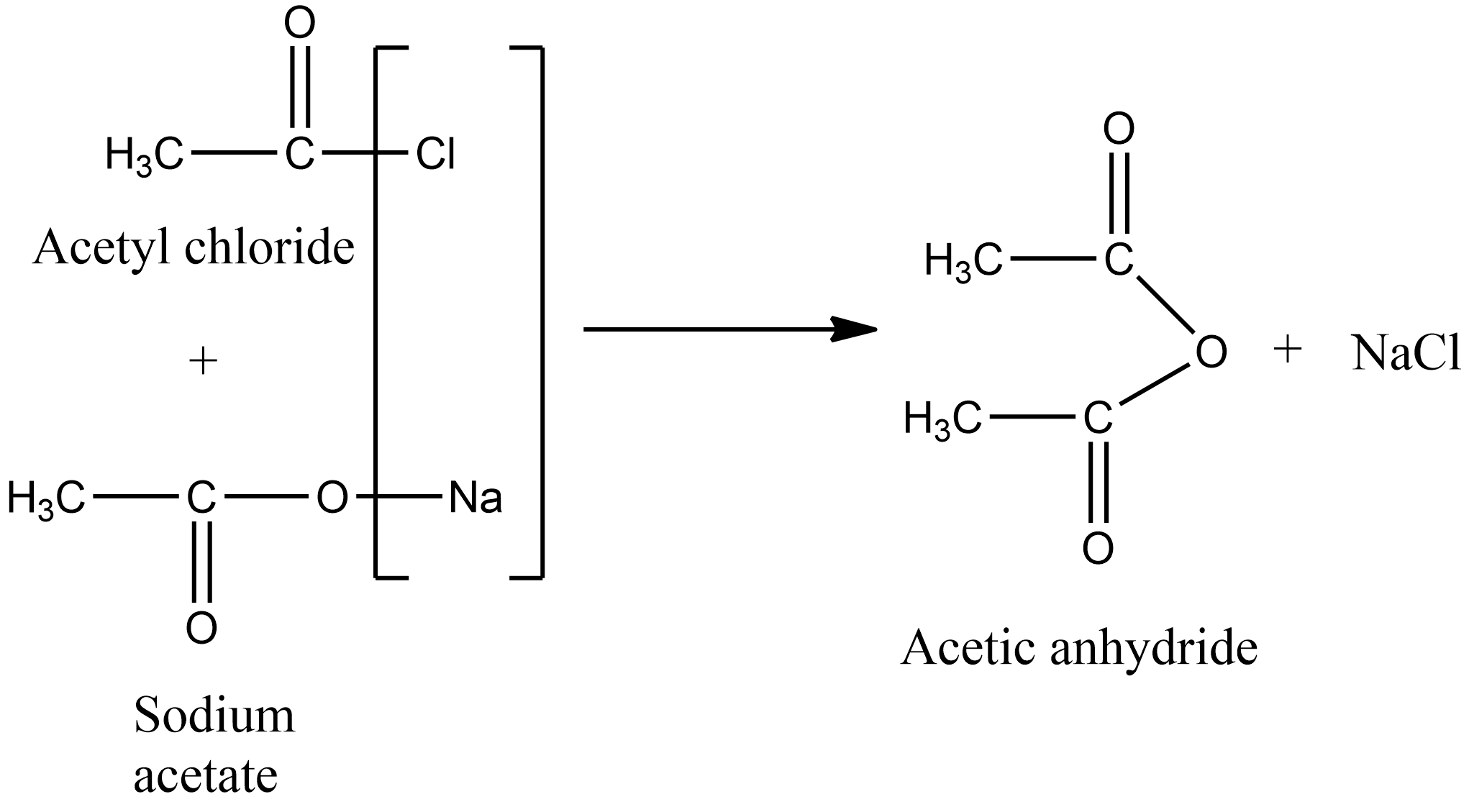
The reaction rate constant of the hydrolysis of acetic acid anhydride has been measured at 5, 10 a nd 20°c. The ketene process for the production of acetic anhydride proceeds in two steps:
A mixture of acetic acid and water is, in this manner, similar to a mixture of ethanol and water. 2 13c nmr spectra give simple patterns.
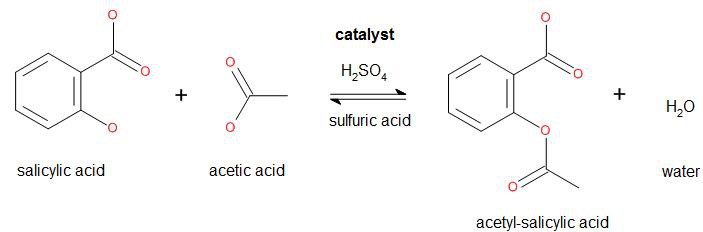
Salicylic acid acetic anhydride acetylsalicylic acid (aspirin) acetic acid preparation of acetylsalicylic acid. Acetic anhydride is most widely used as an agent for acetylation, the addition of an acetyl group to a compound, such as its reaction with ethanolto form ethyl acetate.
The reaction rate constant of the hydrolysis of acetic acid anhydride has been measured at 5, 10 a nd 20°c. The photodecomposition of acetic anhydride has been studied.

Acetic acid and acetic anhydride acetic acid is the chemical compound responsible for the characteristic odor and sour taste of vinegar. Acetic anhydride is most widely used as an agent for acetylation, the addition of an acetyl group to a compound, such as its reaction with ethanolto form ethyl acetate.
As the defining ingredient of vinegar, acetic acid has been produced and used by humans since before the dawn of recorded history. To perform this blank run, inject 1.0 µl of the acetic anhydride.
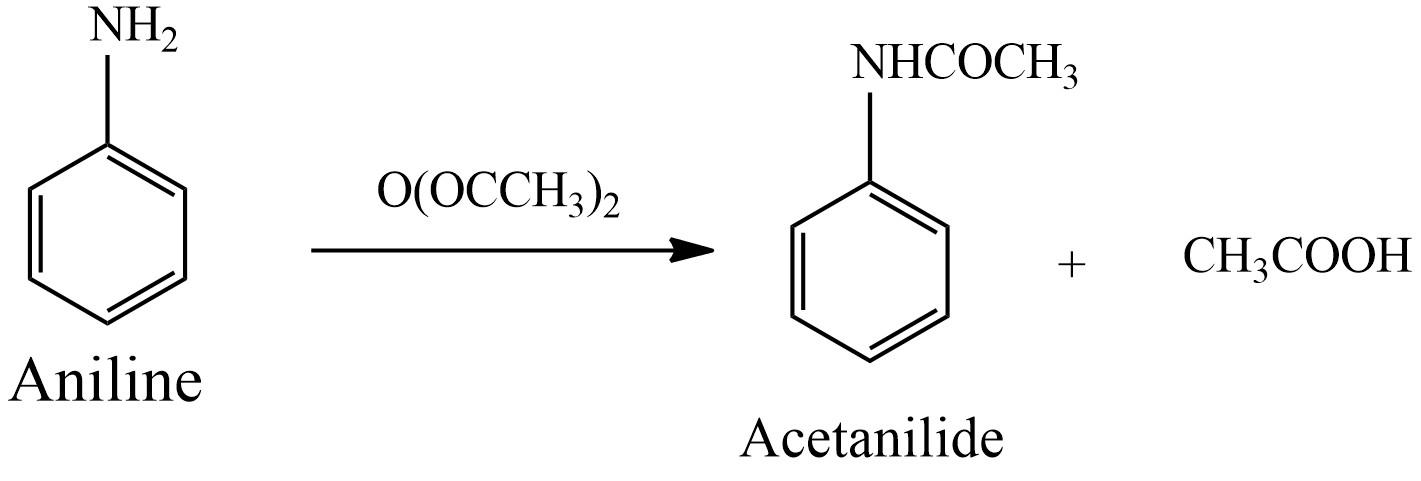
The worldwide production of acetic anhydride is a major application, and uses approximately 25% to 30% of the global production of acetic acid. Acetic anhydride is also a key raw material for aspirin, acetaminophen and.

Acetic acid and acetic anhydride acetic acid is the chemical compound responsible for the characteristic odor and sour taste of vinegar. The key difference between glacial acetic acid and acetic anhydride is that glacial acetic acid is the most concentrated form of acetic acid, whereas acetic anhydride is the dehydrated form of acetic acid.
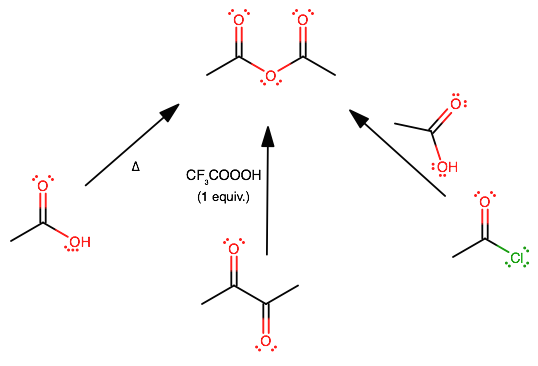
In a distillation column the crude acetic anhydride is distilled and recovered as the bottom product. The thermal cleavage of acetic acid to form ketene and the reaction of ketene with acetic acid.
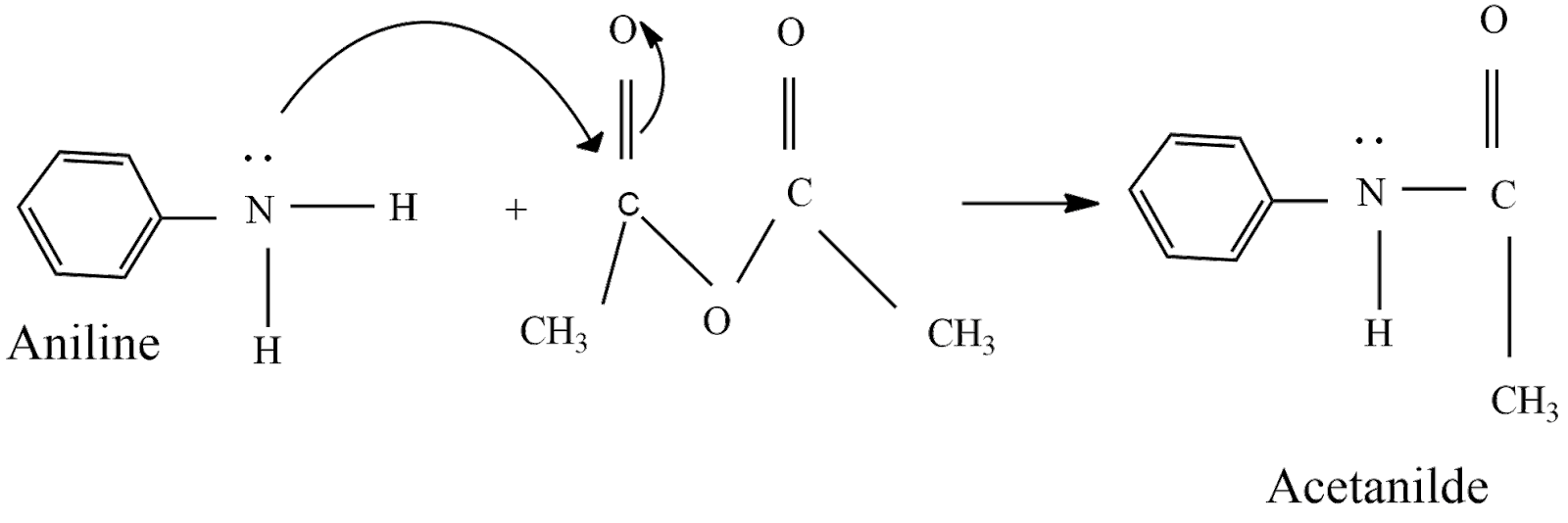
Acetic anhydride is a carboxylic acid anhydride commonly used for the acetylation of amines and alcohols. The largest volume use for acetic anhydride is as a raw material for cellulose acetate fibers and plastics.
The largest volume use for acetic anhydride is as a raw material for cellulose acetate fibers and plastics. In following washing towers the gases are further scrubbed before released to the atmosphere.
It Is Also An Excellent Solvent For Many Cellulose Derivatives And Acetic Acid.Each unique type of carbon atom in a molecule will give a different peak in the spectrum. Acetic anhydride is made from acetic acid. We are the 7th largest manufacturer of acetic anhydride in the world and are 4th largest in terms of merchant sales globally.
As The Defining Ingredient Of Vinegar, Acetic Acid Has Been Produced And Used By Humans Since Before The Dawn Of Recorded History.With pcl5, the reaction is a two step process. The key difference between glacial acetic acid and acetic anhydride is that glacial acetic acid is the most concentrated form of acetic acid, whereas acetic anhydride is the dehydrated form of acetic acid. Here acetic acid acts both as a solvent and as a nucleophile to trap rearranged carbocation.
Acetic Anhydride Is Also A Key Raw Material For Aspirin, Acetaminophen And.Being a carboxylic acid anhydride, acetic anhydride will slowly decay in contact with waterinto acetic acid. This process yields aspirin and acetic acid, which is considered a byproduct of this reaction. Over a period of less than.
As The Defining Ingredient Of Vinegar, Acetic Acid Has Been Produced And Used By Humans Since Before The Dawn Of Recorded History.Its hydrolysis leads to the formation of acetic acid, which is highly. C 4 h 6 o 3. A mixture of acetic acid and water is, in this manner, similar to a mixture of ethanol and water.
This Compound Has A Very Low Density And Is Highly Reactive With Water;Salicylic acid acetic anhydride acetylsalicylic acid (aspirin) acetic acid preparation of acetylsalicylic acid. Accurate adjustment of the electrode is crucial to get precise re sults. Acetic anhydride is most widely used as an agent for acetylation, the addition of an acetyl group to a compound, such as its reaction with ethanolto form ethyl acetate.
Belum ada Komentar untuk "Acetic Acid To Acetic Anhydride"
Posting Komentar